|
In a reversible process, heat absorbed by the system is equal to that lost from the surroundings. = 5.27 cal/deg Entropy change of the universe For example, one mole ice changes into liquid water at 0 ☌ and 1 atmosphere pressure. When heat change occurs at a constant temperature. = 1.18 cal/deg Entropy change at a constant temperature 
When one mole of water is heated reversibly from 27 ☌ to 37 ☌. How to calculate entropy change in thermodynamics? It means entropy remains constant for isolated systems or adiabatic processes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
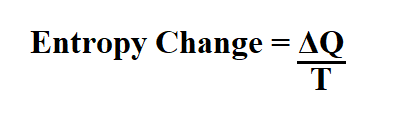

 RSS Feed
RSS Feed
